Calibration your GxP auditor won’t challenge.
MHRA and FDA inspectors expect UKAS-accredited calibration evidence. We provide traceable certificates for temperature, humidity, pressure, flow and air velocity. Certificates returned in 5 days.
Calibration that stands up to MHRA, FDA and GMP audits
Three reasons quality managers at pharmaceutical manufacturers, cold chain operators and life sciences labs use Labcal for their calibration programmes.
UKAS Lab 0625 certificates are accepted by MHRA, FDA and notified body auditors without challenge. Documented uncertainty of measurement, ILAC Mutual Recognition recognition, and traceability chains on every certificate.
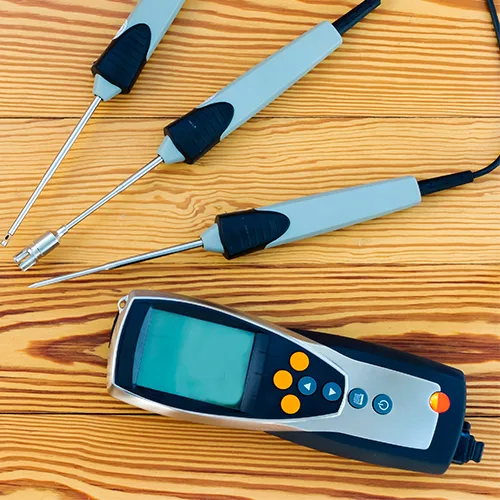
Temperature mapping probes, humidity probes, pressure differentials and air velocity for cleanrooms and passbox testing. The parameters regulators check most in pharma audits are the parameters we’re accredited for.
Stop splitting pharma calibration across three or four suppliers. Labcal covers temperature, humidity, pressure, flow, gas flow and air velocity under one UKAS schedule.
Every calibration under one UKAS schedule
Consolidate your calibration with one accredited lab. No split shipments, no mixed certificate formats, no extra audit complexity.


Around half of the quality managers who enquire with us are leaving another provider. We’ll match your existing scope, parameters and tolerances, then come back with a price and turnaround inside 24 hours. No switching cost, no rework.
What pharmaceutical quality managers ask us
The questions most commonly raised by quality managers, QA leads and validation engineers before they move calibration to Labcal.
Request a calibration quote
Tell us what you need calibrating. We respond to all enquiries within one working day with a price and turnaround time.
We aim to respond within one working day. Average turnaround is 8 days once instruments are received.