UKAS-accredited calibration supporting regulated industries.
ISO 13485 and EU MDR require traceable measurement and calibration processes. Labcal provides UKAS-accredited calibration certificates with traceability and stated measurement uncertainty to support regulated quality systems.
UKAS-accredited calibration for regulated medical manufacturing
Labcal’s UKAS-accredited calibration certificates support traceability and measurement control requirements within ISO 13485 and FDA-regulated quality systems.
Labcal supports customers operating under ISO 13485 and FDA-regulated quality systems with UKAS-accredited calibration services and traceable measurement capability.
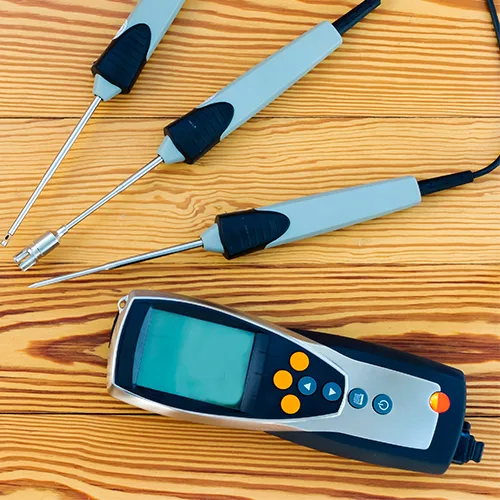
We calibrate the temperature and humidity sensing instruments used within autoclaves, ETO sterilisers and packaging environments, plus flow on process equipment. The measurements your validation protocols depend on, all under one UKAS schedule.
Labcal provides consistent UKAS-accredited calibration services for instruments used in R&D, verification and production environments, supporting traceability throughout the product lifecycle.
Every calibration under one UKAS schedule
Consolidate your calibration with one accredited lab. No split shipments, no mixed certificate formats, no extra audit complexity.


Around half of the quality managers who enquire with us are leaving another provider. We’ll match your existing scope, parameters and tolerances, then come back with a price and turnaround inside 24 hours. No switching cost, no rework.
What medical device quality teams ask us
Practical answers for quality managers, regulatory affairs leads and validation engineers at medical device manufacturers working to ISO 13485, MDR or FDA requirements.
Request a calibration quote
Tell us about your medical device calibration needs. We respond to all enquiries within one working day with a price and turnaround time.
We aim to respond within one working day. Average turnaround is 8 days once instruments are received.